The Evolution of Mohs Surgery
By Perry Robins, MD with Tobechi L. Ebede, MD, and Elizabeth K. Hale, MD
The Single Most Effective Skin Cancer Treatment
It is almost inconceivable how much the treatment of skin cancers has changed in the past 50 years. In the mid-1960s when I was establishing my career in dermatology at New York University, we essentially had only three modes of treatment: curettage and electrodesiccation; standard surgical excision of the tumor, and radiation. The cure rate for these techniques was about 90 percent when removing primary tumors, and only about 50 percent or lower when treating tumors that recurred after the initial treatment.
Today, a far greater variety of therapies exist, from topical medications and cryosurgery (freezing the tumors with liquid nitrogen) to lasers. However, one technique that came of age in the late 60s and 70s stands out above the rest: Mohs surgery is currently the most precise, tissue-sparing method for the treatment of basal cell and squamous cell carcinomas, the most common cancers in the United States.1 The goal of Mohs surgery is complete tumor removal with maximum preservation of healthy tissue. Cure rates are up to 99 percent for primary lesions, and an astonishing 94 percent for recurrences unsuccessfully treated by other methods — impressive results for a technique that was barely known in the medical community half a century ago and aroused outrage when first announced as a skin cancer cure.
The Beginnings: Chemosurgery
In 1933, 23-year-old Frederic Edward Mohs was a research assistant assigned to inject different chemicals into cancerous rat tissues to produce specific reactions. He discovered that one of these chemicals, a zinc chloride solution, could “fix” skin tissue for microscopic study — preserve it without changing the architectural structure of the cells.2 He combined zinc chloride solution with stibnite and sanguinaria canadensis to develop a cohesive paste. When he applied the paste, Mohs found that he could excise the tissue without causing bleeding. He could then prepare frozen sections of the excised tissue, and placed them on slides to be viewed under the microscope. Thus began his groundwork for the technique that today bears his name.
In 1936, after training as a surgeon, Dr. Mohs began performing the procedure, initially dubbed “chemosurgery” (“chemo” referring to the zinc chloride paste), on human skin cancer patients. It was a scrupulous process that could take days. Once clinical examination revealed a potential cancer, he would first apply dichloracetic acid to the area believed to be involved;3 this would allow him to scrape away the protein keratin from the epidermal layer of the skin, so that the zinc chloride paste could penetrate the area. The paste would then be applied, fixing the tissue. A protective dressing would be placed over the treated site to enhance penetration and absorption of the paste.

When adequate fixation was achieved many hours later or by the next day,4 Mohs would remove the dressing and, using a scalpel, would surgically excise a saucer-shaped layer of the fixed tissue. He cut the specimen into sections about 1 cm square in area and 2 mm thick, and sketched a corresponding map of the sections, keeping each section separate and numbered to indicate its location on the map.3 He also painted the adjoining edges of the sections with different dyes to differentiate the superior edges from the inferior. Reference marks were also made on the original surgical site to show exactly where each color-coded section had originated. Thus, any further surgery that might be required would occur only in the part of the surgical site where a cancerous section had been found.
Frozen sections were prepared of the undersides of each excised section, and they were mounted on slides and examined under the microscope. Each slide was labeled to indicate the particular section’s position on the map. If sections showed no evidence of cancer, no further surgery would be required. However, if a section revealed cancer cells, surgery would continue only in its corresponding portion of the surgical site. The zinc chloride paste would be reapplied to that area, and the entire procedure repeated, until no more cancer cells remained in any specimens. Five to seven days later, an “eschar” (a scab on the fixed tissue site) would separate, leaving healthy, healing tissue behind. Surgical repair of the wound could then be performed.2, 3, 5 The technique had many advantages over standard excisional surgery, where the physician simply removes the tumor and a wide extra swath of presumably normal skin around it as a safety margin.6 First and foremost, chemosurgery allowed 100 percent of the cancer to be removed while sparing the most possible tissue. By avoiding overly aggressive surgery and the removal of too much healthy tissue, it produced a much better cosmetic result.

The First Mohs Team at NYU. Right to left: Dr. Perry Robins, Dr. Frederic E. Mohs, Dr. Alfred W. Kopf, Leona Mandell, Dr. Hugh Brown.
Early Struggles
When Dr. Mohs first spoke of chemosurgery at a conference of plastic surgeons, he was greeted with nearly universal dismay and skepticism. When I began to practice the technique in 1965, only one out of every five dermatologists and possibly one of out every 20 physicians were aware that this technique for treating skin cancers even existed.7
In the fall of 1965, I visited Dr. Mohs in Madison, WI, where he was using chemosurgery daily. After five weeks of training, I purchased a “how-to” kit, a jar of zinc chloride paste and Dr. Mohs’ textbook, which I took back to New York. I began to put what he had taught me into practice, but I was initially met with the same kind of resistance he faced. Many of my colleagues considered chemotherapy “black magic,” and pointed out how uncomfortable the zinc chloride was to patients. They believed that dermatologists should not also be surgeons. Some attempts were even made to have me removed from the NYU faculty and expelled from the American Academy of Dermatology (AAD).8
Nonetheless, the potential value of the technique was all too clear to me, and I was convinced that with some additional training, dermatologists could become experts in excising skin cancers with the procedure. After all, we were the physicians best trained to recognize skin cancers, so we should also be the best at removing them. And chemosurgery seemed to guarantee the most success.
Fortunately, a few other physicians agreed with me. At an international meeting, five other Mohs surgeons and I had a founders meeting at a dinner hosted by Dr. Mohs in Munich, Germany. We agreed to form a chemosurgery society and to hold our first meeting at the next annual meeting of the American Academy of Dermatology (AAD), in Chicago. In December of 1965, we indeed held our first meeting in the Palmer House’s “number 10 Wabash Room,” where we continued to meet for years. Only 20 of us attended that first meeting — we were essentially the only physicians practicing the technique in the country.8
In my first year using chemosurgery, I treated 70 patients, and each year that number increased slowly. I found that the technique could also work well in other locations besides the head and face. For example, a few patients were referred to me with skin cancers on their fingers that were scheduled for amputation, and I was able to save the digit. Some also were referred to me with cancers on the penis, and we were able to save them from amputation as well.8
My good friend and colleague Alfred W. Kopf, MD, recognized the value of the technique and felt that doctors practicing it should be well trained. He helped me establish the first-ever dermatologic fellowship for chemosurgery between 1968 and 1969 at NYU.8 We taught the full complement of skills required to perform the technique successfully, including head, neck, and ocular surgery; medical and surgical oncology; gross anatomy; dermatopathology; wound care, and plastic and reconstructive surgery. Dermatologists began coming from around the country and other countries such as Germany and Portugal to train with me, which helped disseminate the technique to Europe and elsewhere. It has been estimated that I or my students trained 40 percent of the current Mohs surgeons worldwide.5, 7, 8, 9
The Fresh-Tissue Technique
Unfortunately, the technique as it first existed had certain drawbacks, above all a procedure time that could span several days, and severe discomfort accompanying the zinc chloride paste application.2, 5, 10
In 1953, Dr. Mohs tried a “fresh-tissue” version of chemosurgery. He was treating a patient with an eyelid basal cell carcinoma, and to avoid irritating the globe of the eye, he skipped the zinc chloride paste. He administered local anesthesia, and without having to wait for a fixative to set, he was able to immediately excise one thin layer of fresh tissue, section it in the usual way, map it, and examine it under the microscope.11 Finding tumor cells at the margins, he was again able to excise another layer of tissue immediately.7, 8, 11, 12
Encouraged, Dr. Mohs began to use this new technique, primarily for skin cancers around the eye. After learning of the technique and excited by the possibilities, dermatologic surgeon Theodore Tromovitch in 1963 began using the fresh-tissue technique on more and more body sites.6,11 In December, 1970 at the annual Chemosurgery Conference, he and Sam Stegman, MD , presented 104 cases of surgery “without using zinc chloride chemical fixative,” with only four recurrences.8,11 He and Dr. Stegman subsequently published landmark papers reporting high cure rates using the new form of chemosurgery, which they called “chemosurgery fresh tissue technique.” In his 8-year retrospective study, he reported a 97.2 percent cure rate for 532 lesions.8,11
I first tried the fresh tissue technique at the start of the 1970’s. In October of 1970, at the annual meeting of the American Academy of Ophthalmology and Otolaryngology (AAOO) in Las Vegas, Rex Amonette, MD, and I presented a number of cases of tumors on or around the eyelids treated with serial excisions of fresh tissue. Like Dr. Mohs, we had skipped the zinc chloride paste for fear of damaging the globe. Following the surgical removal of these eyelid cancers, the wounds healed spontaneously and uneventfully.8
In 1972, I began a study on the effectiveness of the new technique, comparing it with the fixed tissue technique. The first year, I did about a third of my cases with the fresh tissue technique, and two thirds with the fixed. The next year, it was half and half, and the next year I was using the fresh tissue technique two thirds of the time. Though I first doubted the fresh tissue technique, my study showed that the two techniques were equally effective. And the newer technique had many advantages:
1. It eliminated the discomfort of the zinc chloride paste.
2. You could resolve a skin cancer case in one day instead of many days — in fact, you could finish many cases in one day.
3. You didn’t have to wait the 5-7 days for the eschar to separate before beginning wound repairs; you could begin repair and reconstruction the same day.
4. You eliminated the possibility of excessive bleeding when the eschar separated from the healing tissue underneath.8
Since 1974, I have used only the fresh tissue technique.8 In 1980, I published research on 2,900 cases of BCC treated with the fresh-tissue technique, reporting 98.2 percent cure rates for primary BCCs, and 96.6 percent cure rates for recurrent BCCs, significantly exceeding the rates found with standard excision. I also reported similarly improved rates for SCCs. In the years since, the vast majority of Mohs surgeons have used the fresh-tissue approach.8
With the advent of the “fresh tissue” technique, the term “chemosurgery” kept evolving. At first, the two methods were called “chemosurgery fixed tissue technique” and “chemosurgery fresh tissue technique,” but it ultimately seemed absurd to use “chemosurgery” as part of the title if you were no longer using the chemical fixative. Dr. Günter Burg in Munich published a paper calling it “histographic surgery.” Finally, in 1974, Dr. Daniel Jones coined the term “micrographic surgery” to highlight the technique’s use of the microscope and the drawing of tissue maps. At the 1985 annual meeting of the American College of Chemosurgery, the name of the procedure was officially changed to “Mohs micrographic surgery,”5,6 but for brevity’s sake, many just call it Mohs surgery.
Current Practice
Today, there are over 900 members of the American College of Mohs Surgery (ACMS), all of whom have received post-residency fellowship training in Mohs surgery, pathology and reconstructive surgery.14
Currently, Mohs surgery is indicated for basal and squamous cell carcinomas (BCCs and SCCs) with the characteristics listed in Table 1,15,16 and multiple studies have confirmed its superiority over standard excision — in fact, over any other treatment modality — for these purposes, with five-year recurrence rates as low as 1 percent for BCCs and 3-5 percent for SCCs.1,17 Standard excision may be preferable for some small or less aggressive cancers, and for cancers on larger open expansive areas of the body.
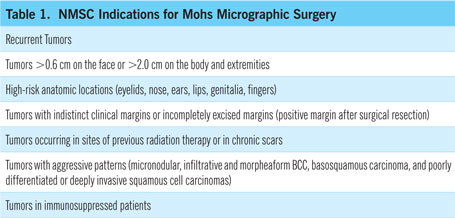
Mohs surgery can also be used to treat other skin cancers, including dermatofibrosarcoma, extramammary Paget’s disease, certain melanomas, Merkel cell carcinoma, sebaceous carcinomas, and microcystic adnexal carcinomas.4, 18
Of Mohs and Melanoma
The use of Mohs surgery to treat melanoma, still relatively new, continues to evolve. For many years, melanoma was not treated with Mohs, because atypical melanocytes (the pigment cells where melanomas can develop) were difficult to assess using frozen sections. More recently, however, special stains called immunostains allow the Mohs surgeon to see possible residual tumor that may not be clearly visible with regular staining.19 For example, pathologists and select Mohs surgeons have begun using the MART-1 (Melanoma-associated Antigen Recognized by T cells) stain to evaluate margins on Mohs frozen sections to diagnose and treat melanoma; MART-1 is especially sensitive and specific for melanocytes.20
To sum up, Mohs surgeons are best equipped to treat skin cancers. They know diseases of the skin, they know dermatopathology, and they are trained to do excisions and repairs. With its unparalleled cure rates, Mohs surgery (also known as Mohs micrographic surgery) is the treatment of choice for select basal cell and squamous cell carcinomas, in addition to other less common skin cancers.
- Tierney EP and Hanke CW. Cost effectiveness of Mohs micrographic surgery: Review of the literature. J Drugs Dermatol 2009; 8:914-922.
- Shriner DL, McCoy K, Goldberg DJ, Wagner RF. Mohs micrographic surgery. J Amer Acad Dermatol 1998; 39:79-97.
- Robins P and Menn H. Chemosurgery in the treatment of skin cancer. Hosp Pract1970; 5:40-50.
- Snow SN, Madjar DD. Mohs surgery in the management of cutaneous malignancies. Clinics in Dermatol 2001; 19:339-47.
- Hanke CW. History of Mohs micrographic surgery. J Drugs Dermatol 2002; 2:169-74.
- Brodland DG, Amonette R, Hanke CW, Robins P. The history and evaluation of Mohs micrographic surgery. Dermatol Surg 2000; 26:303-7.
- Robins P. Chemosurgery: my 15 years of experience. J Dermatol Surg Oncol 1981; 7:779-789.
- Robins P. 44 years in dermatologic surgery: a retrospective. J Drugs Derm June 2009; 8:6:519-25.
- The history of Mohs micrographic surgery. 32nd Annual Meeting of the American College of Mohs Micrographic Surgery. www.medscape.com/viewarticle/403429_2. Accessed March 8, 2011.
- Freiman A, Ting PT. Mohs micrographic surgery: a historical perspective. Dermanities 2003; 1(3) http://www.dermanities.com/detail_article_63.html. Accessed Feb 11, 2011.
- Tromovitch TA, Stegman SJ. Microscopic-controlled excision of cutaneous tumors: chemosurgery, fresh tissue technique. Cancer Feb 1978; 41:653-658.
- Robins P, Albom MJ. Mohs surgery—fresh tissue technique. J Derm Surg 1975; 1:2:37-41.
- Robins P. The benefits of Mohs micrographic surgery. The Skin Cancer Foundation Journal 2004; 22:49.
- American College of Mohs Surgery. www.mohscollege.org/acms.Accessed Nov 28, 2010.
- Drake LA, Dinehart SM, Goltz RW, et al. Guidelines of care for Mohs micrographic surgery. J Am Acad Dermatol 1995; 33:271-278.
- National Comprehensive Cancer Network. Clinical practice guidelines in oncology: basal cell and squamous cell skin cancers. Version 1. 2010. www.nccn.org/professionals/physician_gls/PDF/nmsc.pdf. Accessed Nov 28 2010.
- Arnon O, Papini R, Mamelak AJ, Goldberg LH. Mohs micrographic surgery: current techniques. Israel Med Assn J 2010; 12:431-435.
- Nelson BR, Railan D, Cohen S. Mohs micrographic surgery for nonmelanoma skin cancers. Clin Plas Surg 1997; 24:705-718.
- El Tal AK, Abrou AE, Stiff MA, Mehregan DA. Immunostaining in Mohs micrographic surgery: a review. Dermatol Surg 2010; 36:275-90.
- Cherpelis BS, Glass LF, Ladd S, Fenske NA. Mohs surgery for melanoma insitu: how we do it. J Drugs Dermatol 2010; 9:786-788.
